Ordering & Inventory
The overall aim of the Ordering & Inventory module is threefold:
- to support researchers in their daily work and to increase the awareness with regard to “safe working in the lab”.
- to alleviate the burden of regular accounting to external auditors.
- to do the above in the most “lean” way possible, saving on unnecessary human labour
 Ordering
Ordering
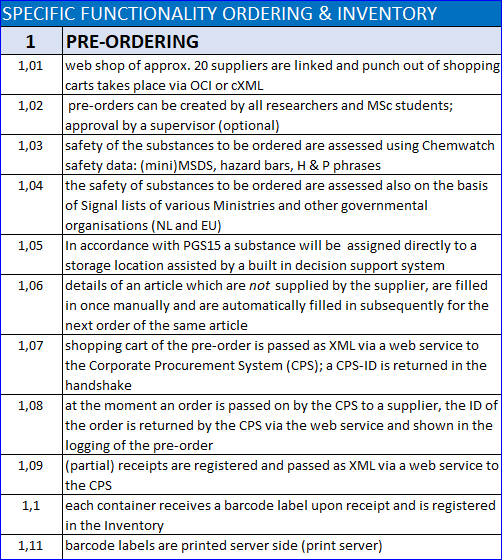
The Lab Servant’s pre-order form is linked to the web shop of preferred suppliers via a web service. The placer of the order fills a shopping cart in the web shop, which is transferred to the order form, where it is merged with financial codes and delivery information of the placer of the order.
As soon as the safety information of the items to order is retrieved from Chemwatch and internal Signal lists of dangerous goods have been consulted, hazard bars, MSDS, ADR class, GHS symbols, H & P-phrases and alerts from the signal list are shown. That way the placer of the order can decide to order the substance taking into regard the safety issues or decide to order an alternative substance, e.g. in the case of CMR and other substances for which the use is discouraged.
After approval by a lab manager the order is transferred to the corporate procurement system for a budget check and e-ordering, e-invoicing and registration in the corporate ERP system.
Suppliers who don’t have a web shop with OCI (yet) can provide a download catalogue, which is integrated in the Lab Servant and offers (almost) the same functionality as a web shop. For other suppliers an open web order form is available to define items to order with their article no., description, price and number to order.
As soon as the articles have been delivered, the goods receipt is registered in the Lab Servant and transferred to the corporate procurement system to release the invoice payment. At the same time a barcode label for each container is printed, the containers are registered in the Lab Servant’s Inventory Module and documents for transport over public roads are generated (ADR-compatible).
Inventory
The purpose of our registration of chemical and biological substances is twofold:
- To know which substances are “in house” and where they are located, saving quests and duplicate orders.
- To be accountable to bodies such as environmental inspection, fire department, labour inspection, customs and security agencies with regard to the nature, quantity and use of existing (risk) substances.
By a live link with Chemwatch, safety data of substances can be displayed directly in the Inventory Module (hazards, precautionary measures, MSDS and mini MSDS, ADR class, CMR properties et cetera). This requires zero maintenance because the CAS no. which is retrieved from the supplier is directly fed into the Chemwatch web service, resulting in the requested data. For mixtures or for substances without a CAS no. the safety data can be entered manually. In such cases, tools are provided to calculate the proper safety data.
Reporting can take place from the grid with substances and containers. The grid can be filterered and the results can be exported to Excel.
In addition a Datamart tool enables a very flexible way of (statistical) reporting. The Dimensions on which to report are selected by drag & drop, as are the Measures which represent the numbers to report. There are various datamarts available to accommodate all reporting needs with a simple “push of the button”. This tool simplifies the reporting obligations to external agencies significantly
On door signs (the Areas, Labs & door signs Module) the amount of hazardous substances per ADR class is displayed, retrieved from the database. The Inventory is further linked to the module Organisation, Users & Roles to provide personalised permissions to access data.
Newly purchased substances enter the Inventory via the module Ordering and are labelled with a unique barcode. Empty containers leave the system by scanning out the barcodes. In this way the inventory is always up-to-date. Additionally, in an annual stock taking process the actual stock is scanned with a barcode reader and compared with the database. Any differences are resolved directly on screen with dedicated action buttons (delete, relocate, restore).
A Warehouse module completes the Inventory. The warehouse comprises 20-200 articles that are kept in stock for same day delivery to the researchers. If stock falls below a threshold re-orders are generated automatically. Derivatives from the warehouse are being used at the laboratory level to keep local stock to the mark automatically, saving on human effort significantly.
Functionality
 Create first-time inventory by scanning supplier’s barcodes, linked to a supplier’s integrated
Create first-time inventory by scanning supplier’s barcodes, linked to a supplier’s integrated
catalogue.- Link container to location: lab – cabinet – shelf – bin.
- Print Lab Servant barcodes to uniquely identify each container and its location and owner.
- Search the database to find a substance needed for an experiment.
- Consult (safety) data of a substance as provided by Chemwatch via a live link with zero maintenance.
- Scan containers to new owner or (temporary) location.
- Scan empty bottles to remove them from the inventory.
- Re-order substances with just a few clicks.
- Print standard overviews for compliance management towards external authorities
For a complete overview of all functionality click the picture
